June 27, 2025
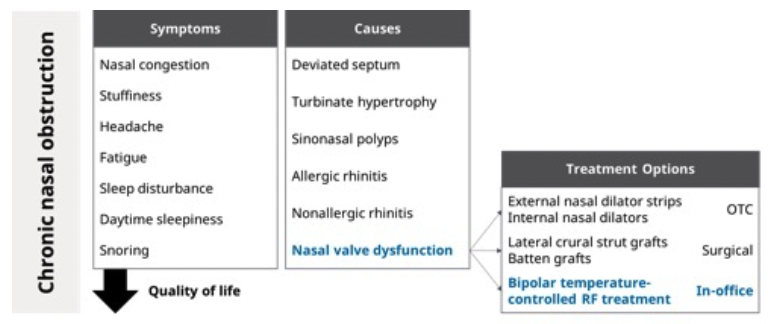
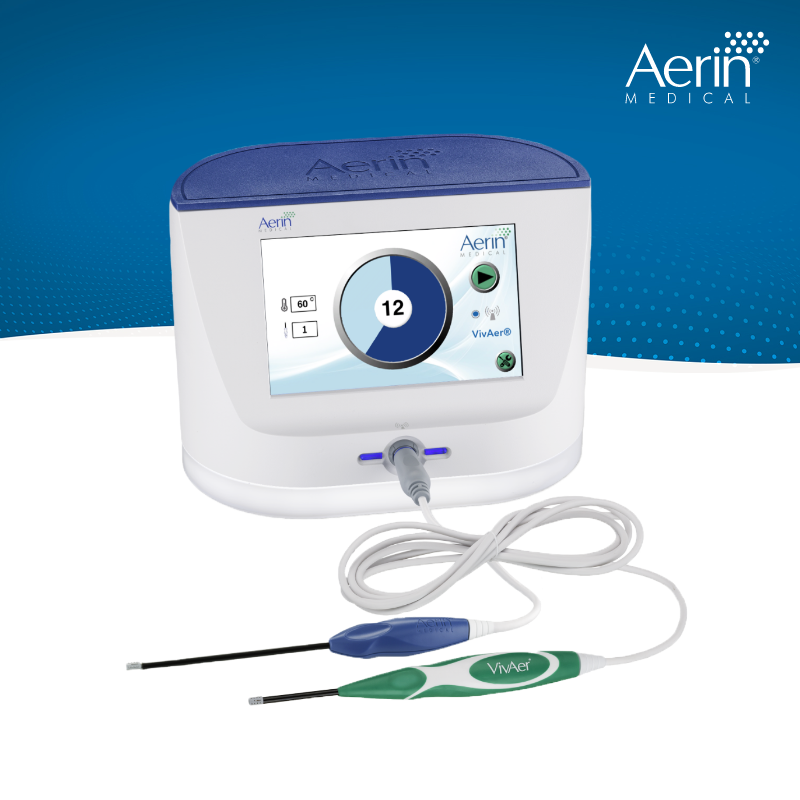
Cigna Enables Coverage for RhinAer®
Starting September 15, over 15 million Cigna members and their healthcare providers will gain access to this clinically proven solution for patients with chronic rhinitis, a persistent inflammatory condition that significantly compromises quality-of-life. Cigna’s policy update highlights the growing recognition of RhinAer’s clinical efficacy and net health benefit as a non-invasive alternative to surgery or long-term, ineffective medication and clinic-based management.
Read More